
Please LIKE this video and SHARE it with your friends!

What functional groups contain carbonyls in the family of amino acids. Topic: Carbonyl Compounds, Organic Chemistry, A Level Chemistry, Singaporeīack to other previous Organic Chemistry Video Lessons.įound this A Level Chemistry video useful? Carbonyls are organic compounds that contain CO bonds that is, they contain.
#Carbonyl functional group how to
The product with the C=N bond is an imine functional group and this gives the distinct orange colour that indicates the presence of carbonyl functional group.įor the detailed step-by-step discussion on how to draw 2,4-DNPH structure and work out the condensation reaction with carbonyl compound, check out this video! To draw the product easily we can point the carbonyl functional group towards the hydrazine functional group, box up water, and join the 2 fragments via a C=N bond. So far we have discussed methods for the formation of the carbon skeleton. The reaction between carbonyl compound and 2,4-DNPH is condensation, where both compounds combine and a small molecule H 2O is eliminated. So we can piece everything together to work out the structure of 2,4-dinitrophenylhydrazine. acetyl group (carbonyl carbon double bonded to oxygen and single bonded to methane) and a chlorine atom. Learn vocabulary, terms, and more with flashcards, games, and other study tools. Key features: all the carbons are sp3-hybridized, only.
You must know the first ten alkanes and their names as they are in the basis of naming and understanding the structure of any other functional groups. They are made of carbons and hydrogens that are only connected with single () bonds. As a result, the carbonyl group is best described as a hybrid of the following resonance structures. Alkanes are the first group of organic compounds. The difference between the electronegativities of carbon and oxygen is large enough to make the CO bond moderately polar.
#Carbonyl functional group full
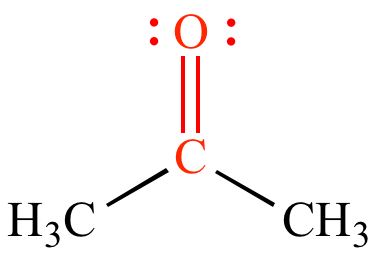
The full name of 2,4-DNPH or Brady's Reagent is 2,4-dinitrophenylhydrazine. Start studying Carbonyl Functional Groups. It is somewhat misleading to write the carbonyl group as a covalent CO double bond. We need to know the structure of 2,4-DNPH for writing balanced equations, and it is easier to remember the full name and structure together. Apparent hygroscopicity decreased with carbon chain length and increased with polar functional groups in the order carboxyl > hydroperoxy > carbonyl. Positive test will be the observation of an orange precipitate. To identify the presence of carbonyl compound we can add 2,4-DNPH at room temperature. All rights reserved.In this video we want to discuss the distinguishing test for carbonyl compounds which include aldehydes and ketones. Copyright © 2012, Columbia University Press. The Columbia Electronic Encyclopedia, 6th ed. Another type of reaction is due to the tendency of the electron-deficient carbon atom of the carbonyl group to partially attract electrons from carbon atoms adjacent to it in the molecule, thus increasing the acidity of hydrogen atoms that are bonded to the adjacent carbon. In ketones, two carbon groups are attached to the carbonyl carbon, while in aldehydes at least one hydrogen is attached to the carbon. The carbon and oxygen in the carbonyl group are sp2-hybridized, with bond angles of 120. One major type of reaction of aldehydes and ketones involves the addition of an electron-rich chemical species to the electron-deficient carbon atom of the carbonyl group. The Carbonyl Group The carbonyl group (CO) is found in aldehydes, ketones, and many other organic functional groups. Because oxygen has the greater affinity for electrons, it acquires a partial negative charge, becoming electron-rich the carbon atom of the carbonyl group thus becomes electron-deficient, acquiring a partial positive charge. The chemical reactivity of aldehydes and ketones is primarily due to the difference in electronegativity between carbon and oxygen. To introduce the chemistry of the carbonyl functional groups. If the carbonyl group is joined only to alkyl groups or aryl groups, the compound is a ketone if it is joined to at least one hydrogen atom, the compound is an aldehyde. A carbonyl group is a functional group featuring a double bond between a carbon atom and an oxygen atom (illustrated below). Carbonyl Chemistry (12 Lectures) Aim of Course To build upon elements of Dr E.H. The carbon atom is joined to the remainder of the molecule by two single bonds or one double bond. Carbonyl group kär´bənĭl, in chemistry, functional group that consists of an oxygen atom joined by a double bond to a carbon atom.
